Kick the tires and light the fires
How Adlershof-based companies deal with the dry spells before regulatory approval for new medicine
Obtaining approval for new products in medical technology can drag on. This can be life-threatening for business founders. How do Adlershof-based companies deal with these dry spells?
Germs in hospital are becoming a serious problem. Particularly in general surgery, we see a rise in cross-infections such as hepatitis, Creutzfeldt-Jakob disease, HIV and the multi-resistant staphylococcus. “This has to do with the use of surgical tools, which can’t be fully sanitised,” says Clemens Frania, CEO of Si Us Instruments GmbH. His business idea was as brilliant as it was obvious: to produce throw-away steel surgical tools, as well as full surgical sets, which are packaged in a cleanroom under sterile conditions. “By using never-before-used instruments, which are thrown away after use, the problem of infections is solved,” Frania explains, who founded his company in 2013. The company’s portfolio comprises 8,000 different products.
“Obtaining approval is an extremely long and difficult process and I’m sure that it is threatening the livelihoods of many start-ups,” says Frania. He was lucky: his matter was settled after a year. Due to stricter rules, medical technology companies today must prepare to wait for two to three years. Without sufficient loans and funding, companies are quickly threatened by insolvency. Frania got through the dry spell by finding a certified partner who acted as the manufacturer, while his company took care of sales. Today, Si Us Instruments delivers its products to the Bundeswehr as well as roughly 150 hospitals.
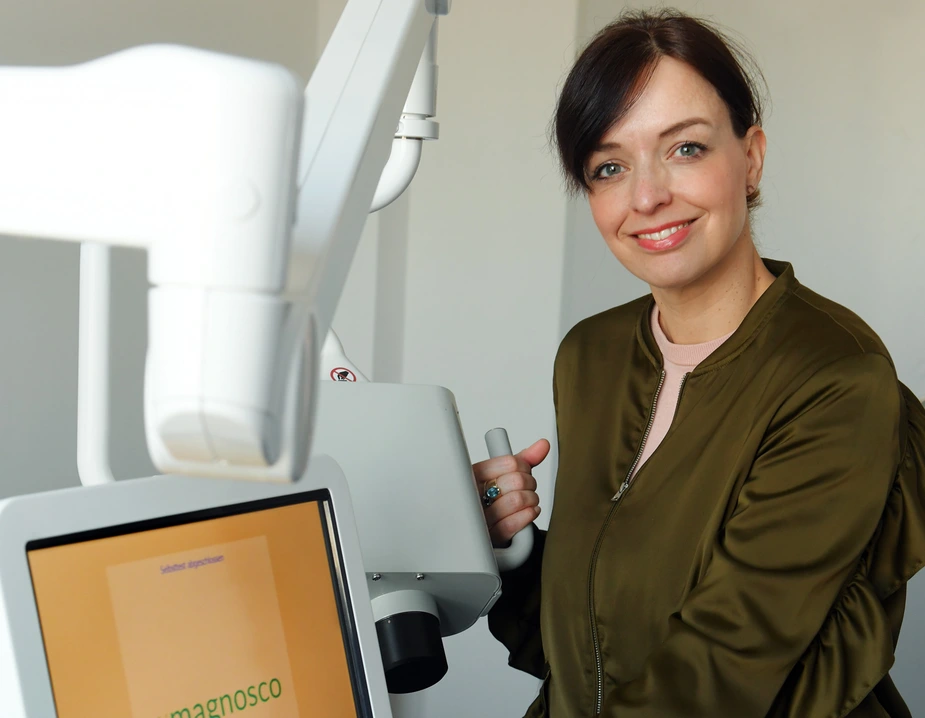
Founded in 2014, the start-up magnosco was extremely lucky with its outside investors: “If you are a company similar to ours, it is important to find investors who understand medical product development and don’t give up easily,” says co-manager Inga Bergen. Magnosco developed the “DermaFC”, a device that detects black skin cancer, which the company loans to doctors for a fee. “The approval process is lengthy but basically predictable,” she says. However, there is a caveat: to get a response from the authorities does not only take time, “you never know when you will get it, which makes it impossible to plan,” says Bergen. This is an obstacle to innovation particularly for small and medium-sized companies seeking medical product approval.
Smarterials Technology, a company founded in October 2016, could also do with more commitment and planning security. The founders developed surgical gloves that prevent needlestick injuries, which they want to put on the market in the fourth quarter of next year. “Multi-layered surgical gloves prevent what is called ‘double gloving’, wearing two pairs of gloves at once,” explains CEO Martin Bothe. “Our goals are to improve infection prevention and perforation indication.” Despite efforts for prevention, needlestick injuries among doctors in Germany amount to half a million every year. Improved structural robustness prevents t perforation of the glove and thus the infection of doctors and patients. “We are currently developing the technology to manufacture such multi-layered gloves here in Adlershof, using existing manufacturing equipment,” says Bothe. When will this innovative product gain approval? He cannot say.
Building a financial buffer is very helpful when going down that path: “Our company is currently being funded by public (ProFIT by the IBB, a Berlin-based business development bank) and private investors,” says Bothe. The start-up can also draw on existing documentation and the help of certified manufactures, which shortens the approval process. The process includes testing the gloves’ shelf life over a period of three years. Comprehensive quality management and extensive technical documentation, which form the backdrop to the required product testing, are constant companions, says Bothe. Frania knows this very well: his company invests 100,000 euros every ear to finance approval for its full range of medical products. This process requires three quality managers who must receive continuous training.
By Chris Löwer for Adlershof Journal
www.si-us-instruments.de
www.magnosco.com
www.smarterials.berlin